So as the Atomic size increase so does Ionization Energy. Ionization energy increases across a period and decreases down the family. 
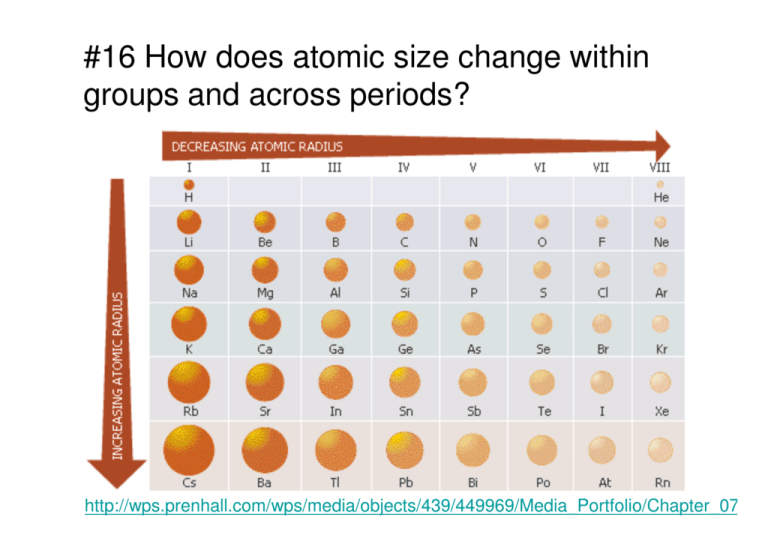
Ionization energy is the energy required to remove the most loosely held electron from a gaseous atom or ion. (a) Atomic size decreases from Na to Cl in the periodic table. Atomic size is the distance from the nucleus to the valence shell. Account for each of the following observations in terms of atomic theory and/or quantum theory. Although there are some reversals in the trend (e.g., see Po in the bottom row), atoms generally get smaller as you go across the periodic table and larger as you go down any one column. AP ® CHEMISTRY 2006 SCORING GUIDELINES (Form B). Seitsonen AP, Schmid M, Varga P (2004) Visualization of atomic processes on. 
This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. J Phys Chem 88:892897 Assmann J, Narkhede V, Khodeir L, Lffler E. Lecture notes atomic radii (size) size of an atom is controlled the size of its orbitals, but orbitals have defined boundary chemists most often use. The third IE, however, is over five times the previous one.\): Comparison of Ionic and Atomic Radius. 1: Atomic Radii Trends on the Periodic Table. Describe and explain the observed trends in atomic size of the elements The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
